cGAS - Novel Therapeutic Target for Uveitis
The cyclic GMP-AMP synthase (cGAS)-stimulator of interferon genes (STING) pathway—a key cytosolic DNA sensor and crucial mediator of inflammation related to infection and cellular stress—has been implicated in the pathogenesis of several ocular diseases, including diabetic retinopathy, age-related macular degeneration, and uveal melanoma.

In a recent study published in Genes & Diseases, researchers from Fudan University, Key Laboratory of Myopia of State Health Ministry, and Peking University People’s Hospital investigated the role of this pathway in mediating inflammatory responses within the EIU model.
Functional role for cGAS in the inflammatory milieu
Using intravitreal lipopolysaccharide (LPS) injections to induce acute ocular inflammation, the authors first characterized the activation of the cGAS–STING signaling cascade in retinal tissue. Molecular analyses demonstrated that the LPS challenge was associated with the enhanced activation of downstream effectors, including TBK1 and IRF3. Concomitantly, increased levels of cytosolic mitochondrial DNA (mtDNA) were observed, suggesting that mitochondrial stress provides the endogenous ligands necessary to amplify inflammatory signaling during endotoxin exposure. Transcriptomic profiling further revealed an enrichment of DNA-sensing and innate immune response pathways, supporting a functional role for cGAS in the inflammatory milieu.
To evaluate the specific contribution of cGAS, the investigators employed cGAS knockout (cGAS⁻/⁻) mice. Genetic ablation of cGAS markedly attenuated the clinical and histopathological manifestations of uveitis. Compared with wild-type controls, cGAS-deficient mice exhibited reduced vitreous haze, diminished retinal vascular leakage, and decreased inflammatory cell infiltration. Furthermore, leukocyte adhesion and macrophage recruitment were significantly reduced, and microglial activation—a key driver of retinal inflammatory amplification—was substantially suppressed. These phenotypic changes correlated with a significant downregulation of NF-κB and MAPK signaling, preserved blood–retinal barrier integrity, and an inhibition of the large-scale upregulation of genes associated with innate immunity and interferon signaling.
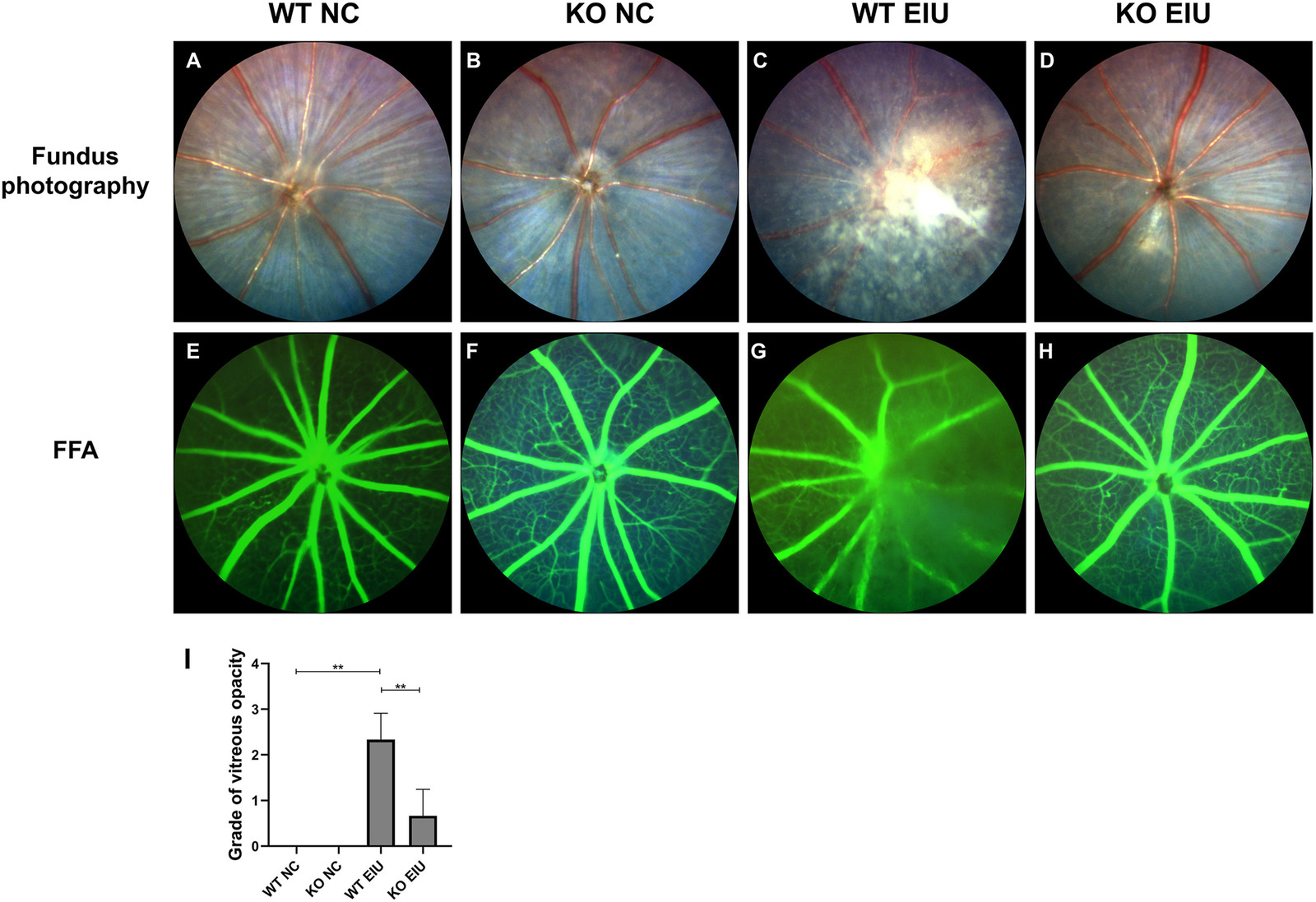
Representative fundus photographs (A–D) and FFA (E–H) of the retinas in the NC and EIU groups. (A) Fundus photograph from the WT NC group. (B) Fundus photograph from the KO NC group. (C) Fundus photograph from the WT EIU group. (D) Fundus photograph from the KO EIU group. (E) FFA from the WT NC group. (F) FFA from the KO NC group. (G) FFA from the WT EIU group. (H) FFA from the KO EIU group. (I) Grade of the severity of vitreous opacity. ∗∗P < 0.01. n = 3 biological replicates per group. FFA, fundus fluorescein angiography; WT, wild type; KO, Cgas knockout; NC, normal control; EIU, endotoxin-induced uveitis.
Reduced inflammation and improved structural preservation
Mechanistically, the study reveals that cGAS senses LPS-induced mtDNA release, catalyzing 2'3'-cGAMP production to activate STING and propagate inflammatory cascades. Inhibiting this axis disrupts the feed-forward loop of pyroptosis, inflammasome activation, and cytokine storms, resulting in reduced inflammation and improved structural preservation. These findings position cGAS inhibitors as promising adjuncts to corticosteroids, particularly for steroid-refractory uveitis.
In conclusion, by delineating the cGAS–STING–TBK1–IRF3 axis, this study identifies cGAS as a pivotal mediator of endotoxin-induced ocular inflammation and highlights it as a high-value therapeutic target for modulating intraocular inflammatory diseases.
Original paper:
Yue Guo, Ruiping Gu, Jiaojiao Wei, Chunhui Jiang, cGAS knockout inhibited endotoxin-induced uveitis in mice, Genes & Diseases, Volume 13, Issue 2,2026, 101786, https://doi.org/10.1016/j.gendis.2025.101786