COVID-19 Virus can breach blood-retinal barrier
The blood-retinal barrier is designed to protect our vision from infections by preventing microbial pathogens from reaching the retina. But researchers have discovered that that SARS-CoV-2 can breach this protective retinal barrier with potential long-term consequences in the eye.
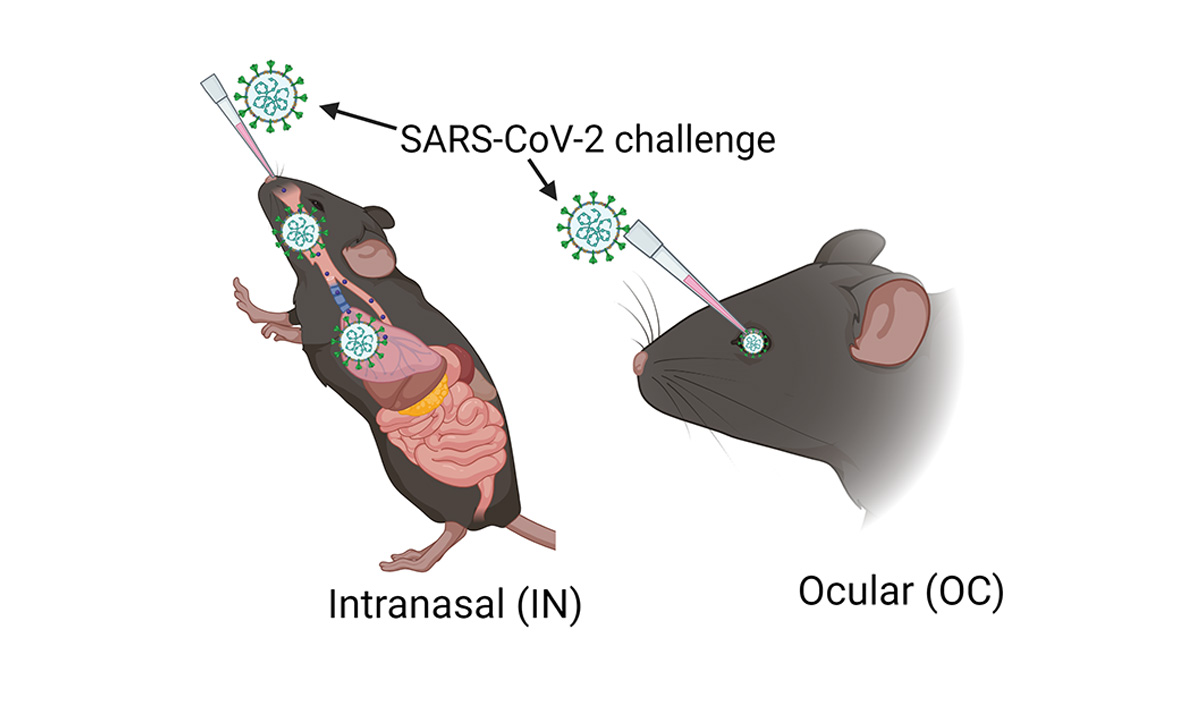
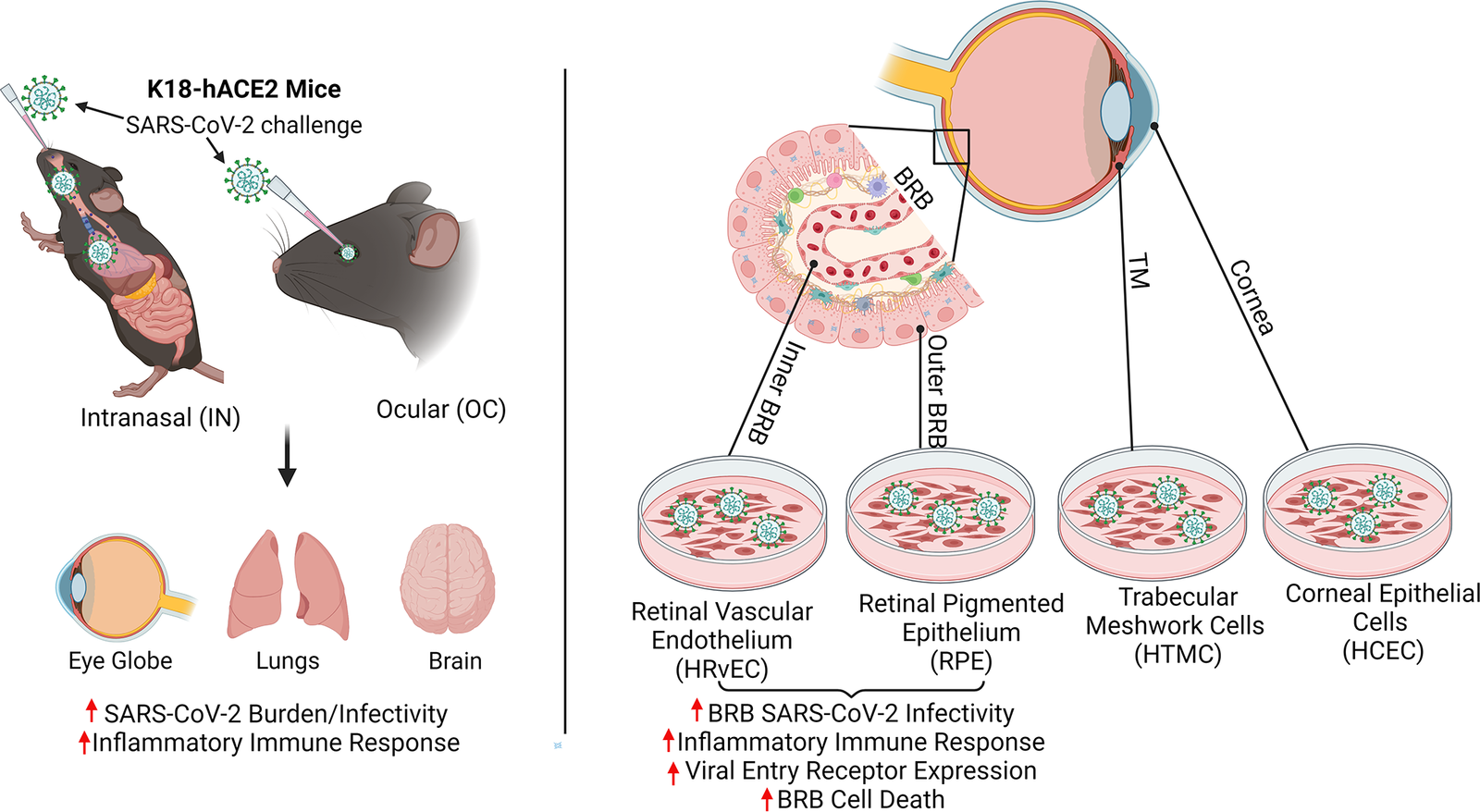
Pawan Kumar Singh, PhD, an assistant professor of ophthalmology at the University of Missouri School of Medicine, leads a team researching new ways to prevent and treat ocular infectious diseases. Using a humanized ACE2 mice model, the team found that SARS-CoV-2, the virus that causes COVID-19, can infect the inside of the eyes even when the virus doesn’t enter the body through the surface of the eyes. Instead, they found that when viruses enter the body through inhalation, it not only infects organs like lungs, but also reaches highly protected organs like eyes through the blood-retinal barrier by infecting the cells lining this barrier.
“This finding is important as we increase our understanding of the long-term effects of SARS-CoV-2 infection,” said Singh. “Earlier, researchers were primarily focused on the ocular surface exposure of the virus. However, our findings reveal that SARS-CoV-2 not only reaches the eye during systemic infection but induces a hyperinflammatory response in the retina and causes cell death in the blood-retinal barrier. The longer viral remnants remain in the eye, the risk of damage to the retina and visual function increases.”
Singh also discovered that extended presence of SARS-CoV-2 spike antigen can cause retinal microaneurysm, retinal artery and vein occlusion, and vascular leakage.
“For those who have been diagnosed with COVID-19, we recommend you ask your ophthalmologist to check for signs of pathological changes to the retina,” Singh said. “Even those who were asymptomatic could suffer from damage in the eyes over time because of COVID-19 associated complications.”
Intranasal exposure of SARS-CoV-2 causes ocular tropism and induction of hyperinflammatory response in the retina through BRB infection, whereas ocular exposure does not cause lung infection and moribund illnesses in K18-hACE2 mice despite the extended presence of viral remnants in various ocular tissues. Cells lining the BRB, outer BRB: RPE, and inner BRB: HRvEC are highly permissive to SARS-CoV-2 infection, whereas corneal epithelial cells are comparatively resistant to infection.
While viruses and bacteria have been found to breach the blood-retinal-barrier in immunocompromised people, this research is the first to suggest that the virus that causes COVID-19 could breach the barrier even in otherwise healthy individuals, leading to an infection that manifests inside the eye itself. Immunocompromised patients or those with hypertension or diabetes may experience worse outcomes if they remain undiagnosed for COVID-19 associated ocular symptoms.
“Now that we know the risk of COVID-19 to the retina, our goal is to better understand the cellular and molecular mechanisms of how this virus breaches the blood-retinal barrier and associated pathological consequences in hopes of informing development of therapies to prevent and treat COVID-19 induced eye complications before a patient’s vision is compromised,” Singh said.
This groundbreaking study entitled “SARS-CoV-2 infects cells lining the blood-retinal barrier and induces a hyperinflammatory immune response in the retina via systemic exposure” was recently published in PLOS Pathogens. In addition to Singh, the research team from the University of Missouri School of Medicine included Vaishnavi Balendiran, MD, vitreoretinal surgery fellow; Monu Monu and Faraz Ahmad, post-doctoral fellows in the Department of Ophthalmology; and Rachel M. Olson, PhD, Chief Scientific Officer, Laboratory for Infectious Disease Research at the College of Veterinary Medicine.
This research was supported through fundings from the University of Missouri and the National Institutes of Health (NIH)/National Eye Institute (NEI).
Source: University of Missouri School of Medicine